MRA
Showing all 9 results
-

CH-REP Manual for Switzerland
Conformity without a certified QMS.
2'000 CHF plus VATSelect options This product has multiple variants. The options may be chosen on the product page -

Checklist for label
for medical devices in EU and Switzerland.
175 CHF plus VATSelect options This product has multiple variants. The options may be chosen on the product page -

EU-Konformitätserklärung (DoC)
Checkliste zur Prüfung der DoC.
50 CHF plus VATAdd to cart -

Importer Manual for Switzerland
Conformity without a certified QMS.
2'000 CHF plus VATSelect options This product has multiple variants. The options may be chosen on the product page -

IVDR Review
Support checking your SOPs against the IVDR.
Read more -

MDR Review
Support checking your SOPs against the MDR.
Read more -


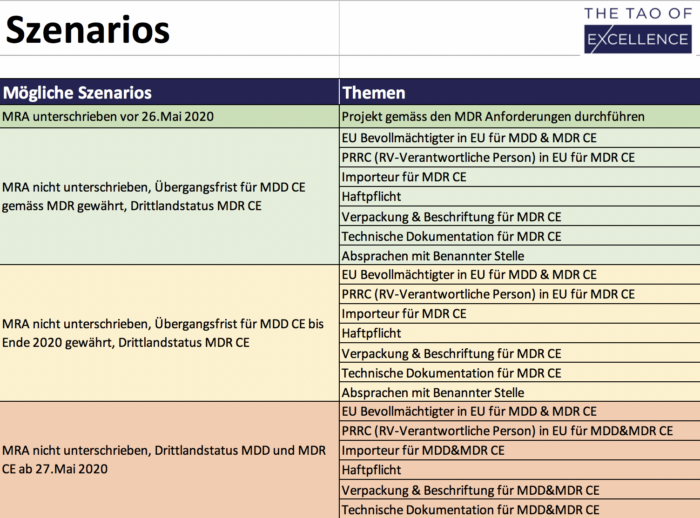
MRA Solutions Finder
Tool to optimize preparation for a missing MRA.
600 CHF plus VATAdd to cart -

RA / QA Membership
Regulatory and quality management support.
Read more -

Regulatory document matrix for medical devices
For the EU and Swiss market
100 CHF plus VATAdd to cart