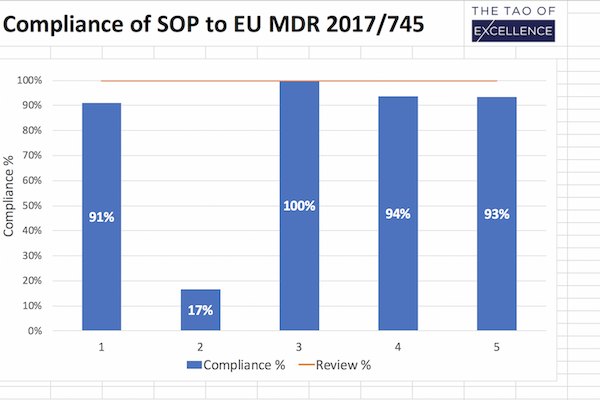
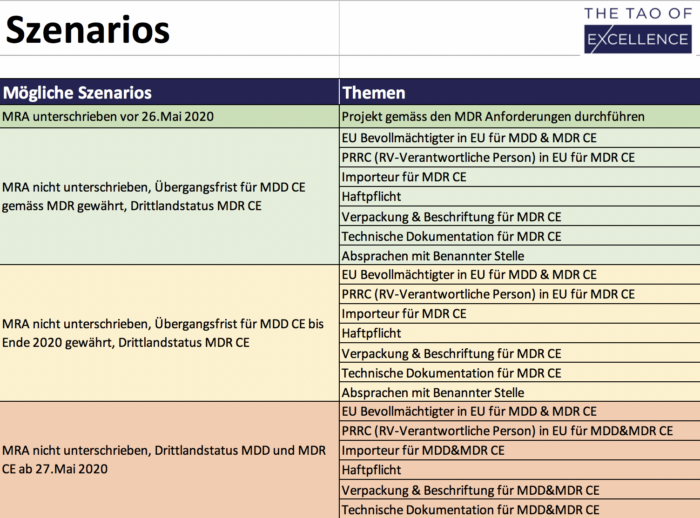
MDR Review Support checking your SOPs against the MDR.
Request a quote
- Complete service that will ensure your SOPs comply to the MDR and standards you require.
- Checking bilingual documents in German and English.
- MDR review report in an Excel document with clear visuals and compliance markers to track and review updates.
- Regulatory and quality management experts that will check the full scope of respective requirements.
- Experienced knowledge and latest insights into requirements for MDR, ISO GCP, ISO 14971, PMS, QMS, ISO 13485, MDSAP, UDI, and more.
Description
Our regulatory and quality management experts will review your standard operating procedures (SOPs) to make sure they comply to the new Medical Device Regulation (EU) 2017/745 (MDR) and any related standards or regulation. As SOPs often involve multiple people and departments it can be helpful to bring in a fresh pair of eyes to review all requirements from start to finish. This service will analyse every aspect of an SOP in relation to the MDR, wider regulation and the standards you require – spanning departments, standards, drafts and documents that can include Good Clinical Practices (ISO GCP), Risk Management (ISO 14971), Post-Market Surveillance (PMS), Quality Management Systems (QMS, ISO 13485, MDSAP), and Unique Device Identifier (UDI), and more.
To request a quote please use the contact form to send us:
- Your contact details
- The number of documents and pages you would like checked.
- The regulation(s) and standard(s) your SOP(s) should be checked against (e.g. ISO 13485:2016).
Request a Quote
You may also like…
-


MDR Risk Management
Gap analysis tool for risk management compliance.
175 CHF plus VATAdd to cart -

RA / QA Membership
Regulatory and quality management support.
Read more -

MDR UDI
Gap analysis tool for UDI compliance.
155 CHF plus VATAdd to cart -

MDR Clinical Evaluation
Gap analysis tool for GCP compliance.
190 CHF plus VATAdd to cart