“We had a very successful audit… Only one deviation (and one really be sought for). Thank you for handing over such a solid system.”
Quality Manager, SME
Our Key Industry Focus
Medical Devices
Recent and steep increases in regulatory requirements is reshaping the medical device industry. Compliance has never been so complex or costly. Ensure that you are properly informed and prepared for the new regulations such as the European Medical Device Regulations (MDR) and Medical Device Single Audit Program (MDSAP).
Combination Products
Technology is progressing at an unprecedented rate. Novel and innovative products require a more integrated approach to process development. Secure your compliance and the sustainability of your products and processes.
Pharmaceuticals
Complex processes within a highly regulated environment make it increasingly difficult to implement improvement or innovation. Reach your project objectives and optimize your Quality Management System.
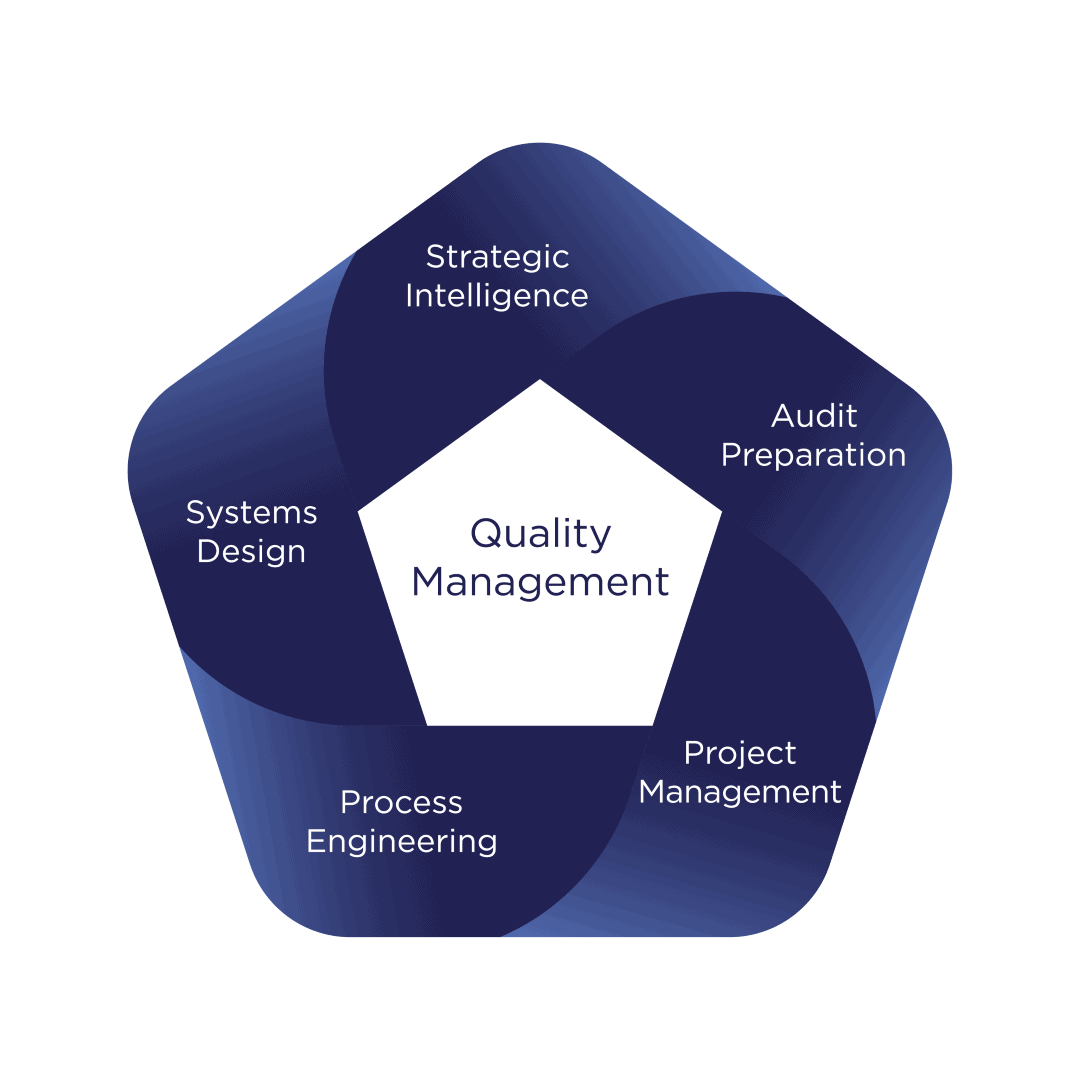
We are here for your quality management and smart process design.
What we do best
-
Swift Solutions
Long-term engagement, specialist expertise and hands-on experience enable us to understand and evaluate your organization and its requirements quickly, allowing us to propose relevant and realistic measures and solutions.
-
Team Integration
We understand that developing and supporting your team is essential to our success.
-
Sustainable Implementation
Your long-term benefit and self-sustainable process design is central to our motivation and approach.
“…they were patient and very present and had the ability to focus on the essential and settle quickly into our team… they got straight into the relevant issues, assessed where we actually were and from there took a step by step approach to reach our goal.”
Employee, SME
Our in-depth understanding of the current regulatory landscape, specialist expertise and state-of-the-art business strategies enable us to deliver a tailor-made service with commercially sustainable quality management.
Our Customers

Large enterprises
“Get ready for the future”
- Strategic advice
- Digital projects
- Project management
- Improvement projects
- Innovation management
- MDR and IVDR certification
- MDSAP certification

SMEs
“Healthy and sustainable growth”
- Sustaining market access
- Interim Management
- ISO9001 and ISO13485 certification
- Business development
- Strategic advice
- MDR and IVDR certification
- MDSAP certification

Start ups
“Secure compliance from the start”
- Complete Quality Management System (QMS) setup
- Reliable contact and support for all quality related topics
- ISO9001 and ISO13485 certification
- MDR and IVDR certification
- MDSAP certification
“We were more than satisfied with how our project goals and your additional services were delivered.”
Supply Chain Engineer, Large Company
“I would like to thank you once again for your coaching on this successful project.”
Team Leader, SME
“What I found very good is that you tackled the whole thing systematically and we really analysed all factors step by step together. As the project progressed, I became more and more confident using the methods you taught me.”
Employee, SME